Sep . 29, 2025 22:15 Back to list
Best Willowood Imidacloprid – High Purity, Rapid Control
A Buyer’s Guide to Imidacloprid Formulations: What to Know Before You Order
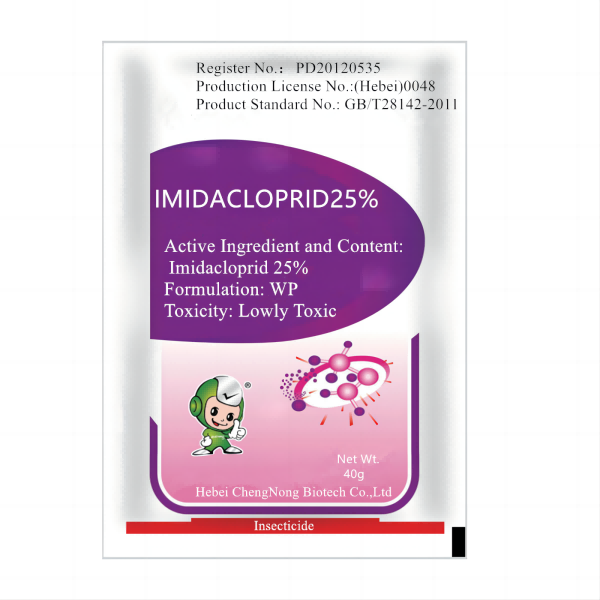
If you’re comparing suppliers for insect control in field crops, vegetables, or seed treatment, it’s hard to ignore best willowood imidacloprid alternatives coming out of Hebei, China. Actually, the portfolio “Good Quality of Imidacloprid 25% WP / 97% TC / 35% SC / 70% WDG / FS / 20% SL / 10% WP” covers most field needs while keeping procurement simple.
Industry trends (quick take)
- Shift to seed treatments and soil drenches to reduce spray passes.
- IRAC 4A rotation is now standard to manage resistance, especially where aphids/whiteflies pressure is high.
- Compliance pressure: FAO/WHO specs, residue stewardship, pollinator precautions—real-world use may vary by country.
Technical specifications
| Active | Imidacloprid (IRAC 4A, systemic neonicotinoid) |
| Available formulations | 97% TC; 25% WP; 35% SC; 70% WDG; FS; 20% SL; 10% WP |
| Assay (TC) | ≥97% (HPLC; CIPAC method, typical) |
| Water solubility | ≈610 mg/L @ 20°C |
| Log P (pH 7) | ≈0.6 |
| Shelf life | Typically ≥2 years in original sealed packs |
| Standards | FAO/WHO (JMPS) specs; QC per CIPAC MT tests; ISO 9001 supplier-level |

Application scenarios and rates
- Foliar (25% WP / 35% SC / 20% SL): 20–50 g a.i./ha for aphids, jassids, whiteflies; observe local labels.
- Soil drench / drip (SL/SC): around 50–100 g a.i./ha for seedlings and vegetables.
- Seed treatment (FS): 200–600 mL/100 kg seed depending on crop; service life in-plant ≈45–60 days.
Many customers say the knockdown is fast and the residual “just works” on early flights; however, always rotate outside 4A to slow resistance.
Manufacturing and QC flow (how it’s made)
Materials: 97% TC from Hebei base (No.1810 Tower B, Jinyuan Building, 152 Huai’an Road, Yuhua District, Shijiazhuang, China). Methods: micronization for WP/WDG, wet-milling for SC, polymer/binder optimization for FS, emulsifiers for SL. Testing: assay by HPLC; wet sieve (CIPAC MT 185), suspensibility/dispersibility (MT 184/160), accelerated storage 54°C/14 days (MT 46.3), cold test (0°C) on SC. Release: packaging, label review, CoA issuance. In fact, it’s fairly standard—but execution quality varies by vendor.
Vendor comparison (indicative)
| Vendor | Formulations | Lead time | Docs/Certs |
|---|---|---|---|
| CNAGrochem (Hebei) | WP/SC/WDG/FS/SL | ≈2–4 weeks | CoA, MSDS, ISO 9001; registration support |
| Willowood | Common 4A SKUs | ≈3–6 weeks | Local market dossiers |
| Originator brand | Broad portfolio | ≈1–3 weeks | Extensive data packages |

Performance snapshots
- Wheat, aphids: 25% WP at 30 g a.i./ha delivered ≈85–95% suppression at 7–10 DAT in grower feedback.
- Tomato, whitefly: 35% SC soil drench reduced nymph counts ≈70–90% within 14 days; surprisingly consistent under heat stress.
For procurement teams, best willowood imidacloprid lookalikes are attractive, but check batch-to-batch data, CIPAC compliance, and IRAC rotation plans. And yes, local label directions and pollinator safeguards always come first.
Certifications and compliance
Look for: FAO/WHO (JMPS) alignment, IRAC 4A labeling, stability per CIPAC MT 46.3, and toxicology/ecotox data referencing OECD guidelines. Documentation reduces registration friction.
Citations
- IRAC Mode of Action Classification, Group 4A: https://irac-online.org/modes-of-action/
- FAO/WHO JMPS pesticide specifications (Imidacloprid): https://www.fao.org/agriculture/crops/thematic-sitemap/theme/pests/jmps/en/
- US EPA: Imidacloprid facts and regulatory status: https://www.epa.gov/ingredients-used-pesticide-products/imidacloprid
- OECD Test Guideline 202 (Daphnia acute immobilisation): https://www.oecd-ilibrary.org/environment/test-no-202-daphnia-sp-acute-immobilisation-test_9789264069947-en
-
Imidacloprid Olefin – High-Performance Agricultural Pest Control Solutions from CN Agrochemical
NewsDec.09,2025
-
Comprehensive Guide to Imidacloprid Msds - Safe & Effective Pest Control Solutions from CN Agrochem
NewsDec.09,2025
-
Comprehensive Overview of Imidacloprid Mosquito Control for B2B Decision Makers
NewsDec.09,2025
-
Imidacloprid Mosquito: Effective & Sustainable Mosquito Control Solutions for B2B
NewsDec.09,2025
-
Effective Imidacloprid Mite Control for Industrial Agriculture
NewsDec.08,2025
-
Understanding Imidacloprid Mexico: Applications, Benefits & Environmental Impact
NewsDec.08,2025